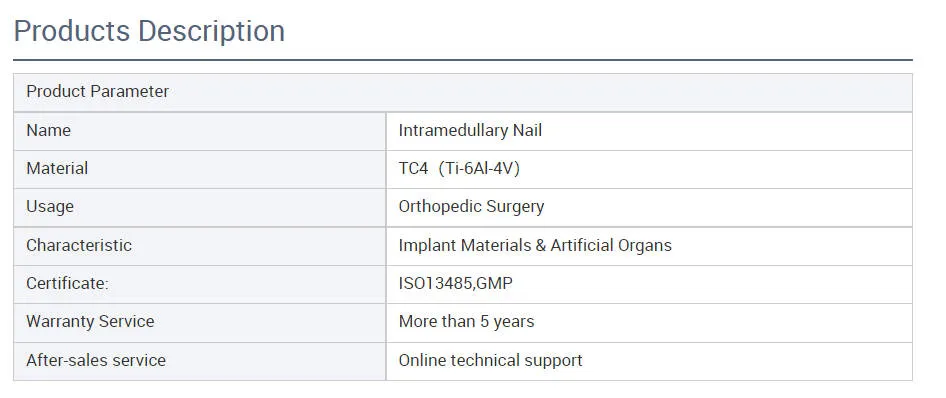
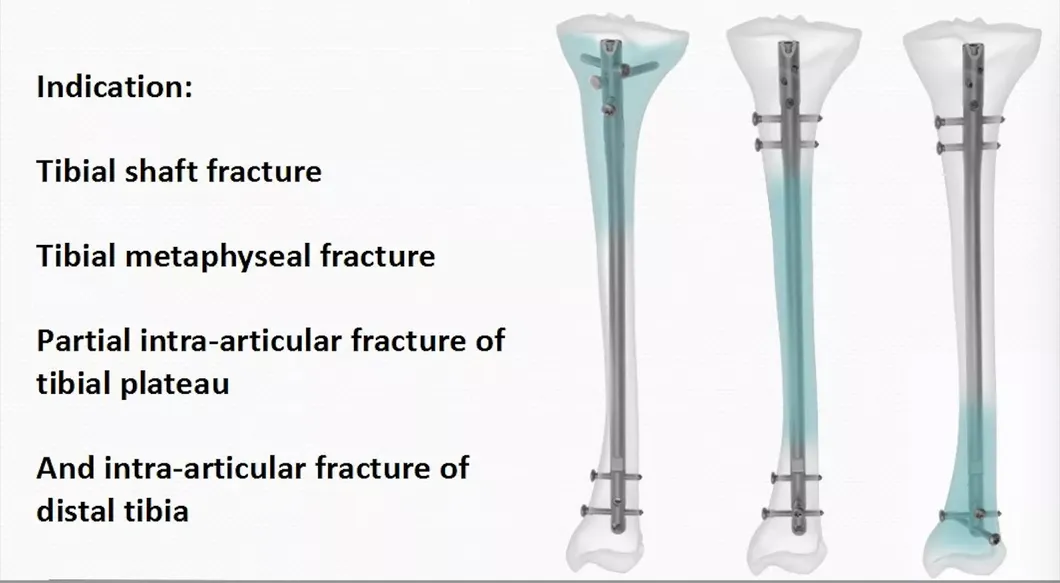
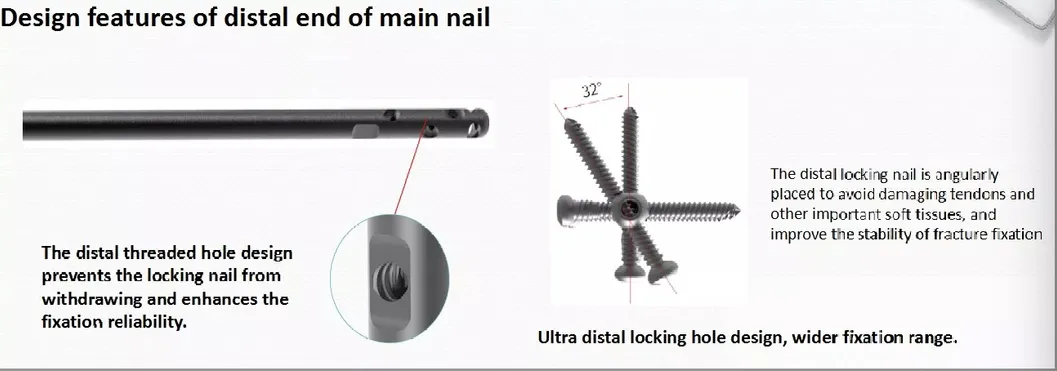
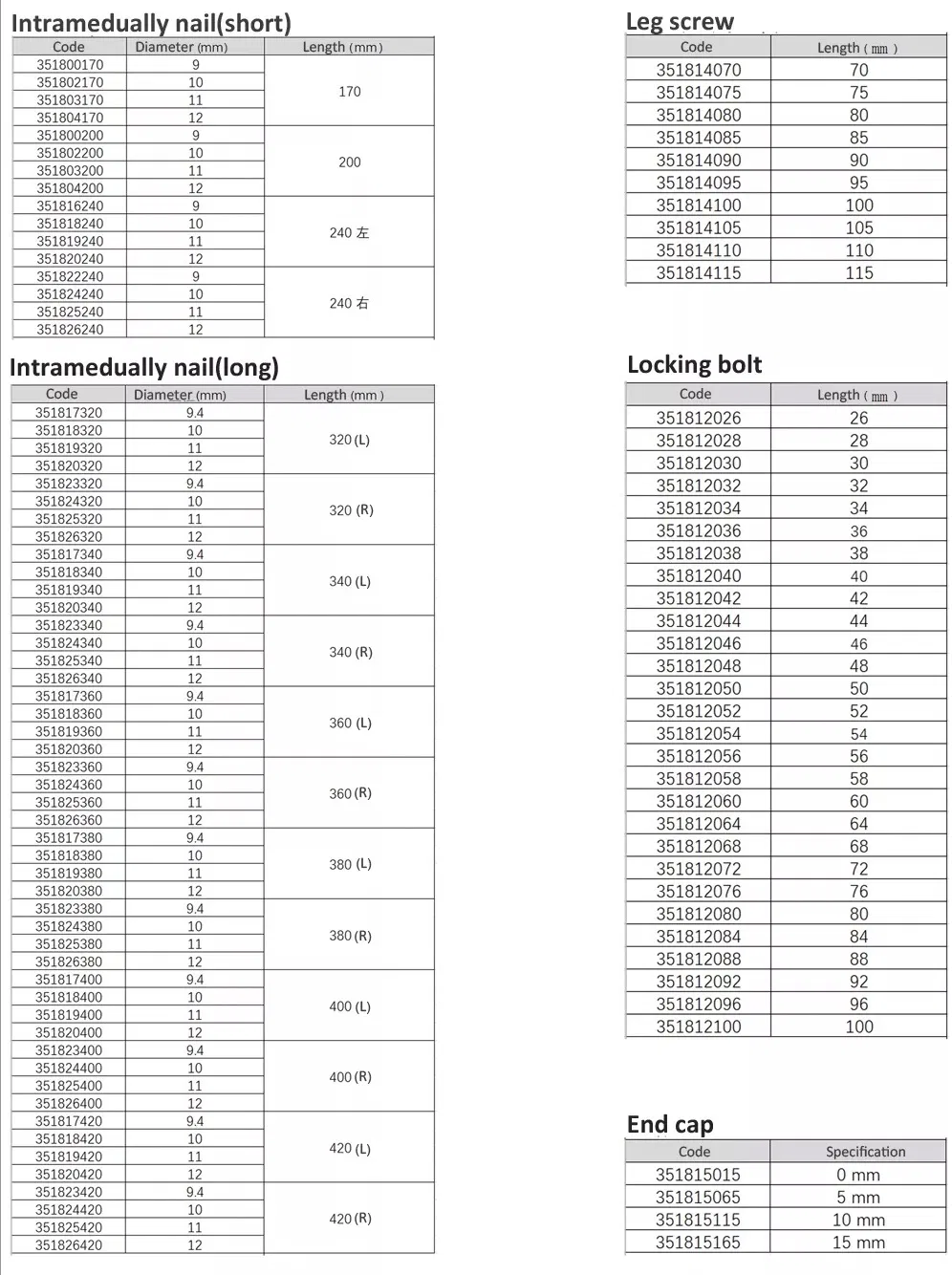
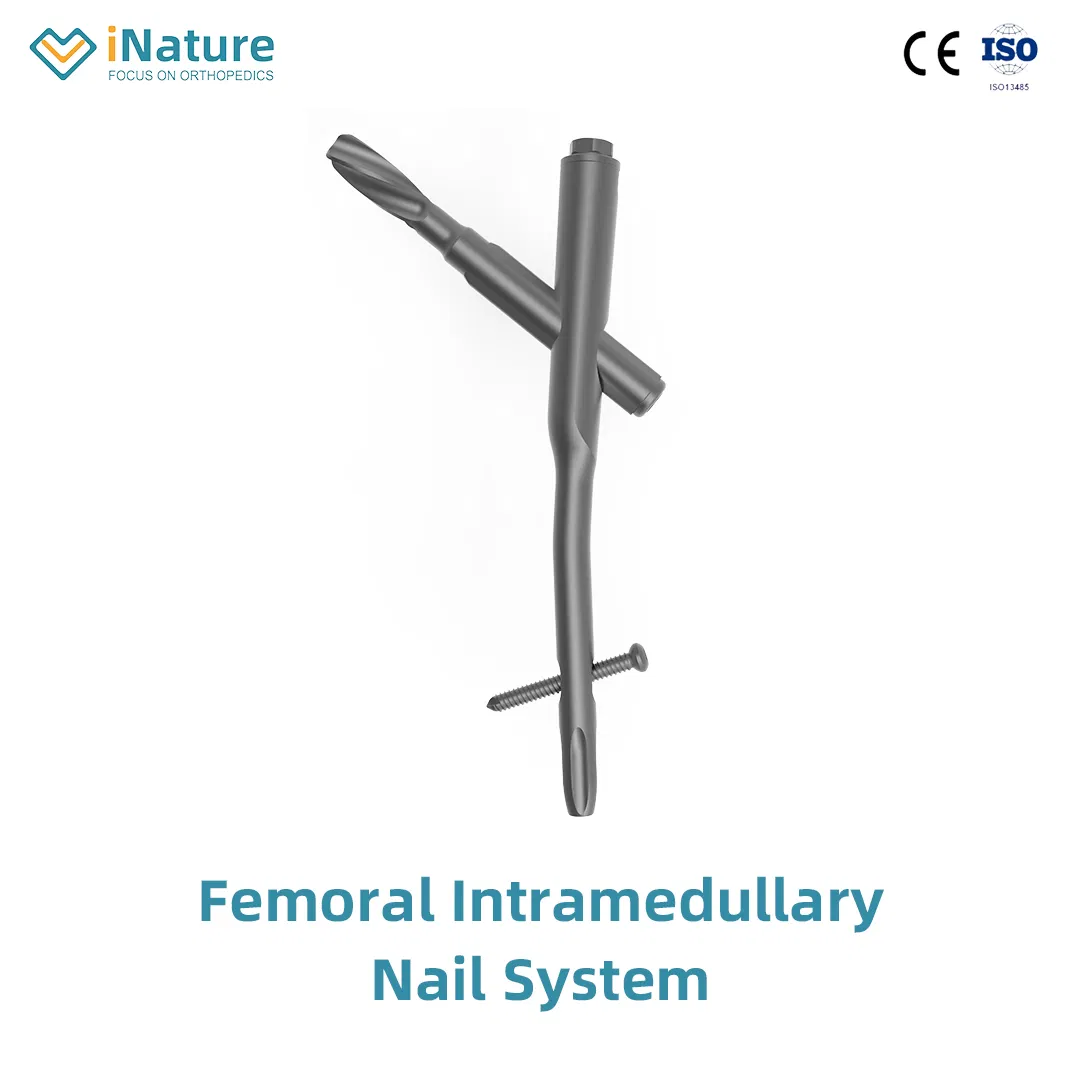
Product Features
✔ Material: High-grade Titanium
✔ Surface Finished: Oxidation / Milling for Titanium
✔ The medial-lateral angle of 5° allows insertion at the tip of the greater trochanter
✔ Lateral flatten design eases insertion and reduces stress to the lateral cortical bone
✔ Elastic groove tip design reduces concentration stress
✔ 240mm distal curved design prevents nail tip against cortical bone, reducing resistance and pain
✔ Lag screw insertion provides excellent compaction for osteoporotic patients
✔ Double thread locking bolt design to reduce operation time
❓ Frequently Asked Questions
1. What kind of guarantee do you provide for these orthopedic implants?
We offer "3 Guarantees": A one-month return/exchange for quality issues, a one-year free repair service, and lifetime technical support and maintenance after the warranty period.
2. Are the prices listed on the website final?
All prices are for reference only. The exact price will be provided based on the latest quotation, order quantity, and specific product requirements.
3. How do you ensure product quality despite not being a "big name" brand?
We focus on a "Win-win" market strategy where quality is equal to the price charged. We compete through rigorous quality control and material standards (Pure Titanium) rather than just low pricing.
4. What material is used for the Intramedullary Nail System?
The system is primarily manufactured using Pure Titanium, which offers excellent biocompatibility and strength for trauma and fracture fixation.
5. Do these products have the necessary medical certifications?
Yes, all products are manufactured under Class III instrument classification and hold CE and ISO 13485 certifications.
6. Is online technical support available for surgical questions?
Yes, we supply timeless maintenance service and our engineers are available to answer any technical questions regarding the product's application and solutions.




 Tandir Orthopedic Implants
Tandir Orthopedic Implants