



1 / 5
 Tandir Orthopedic Implants
Tandir Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |
Key Features and Benefits:

| Product | Size / Length (mm) | Material |
|---|---|---|
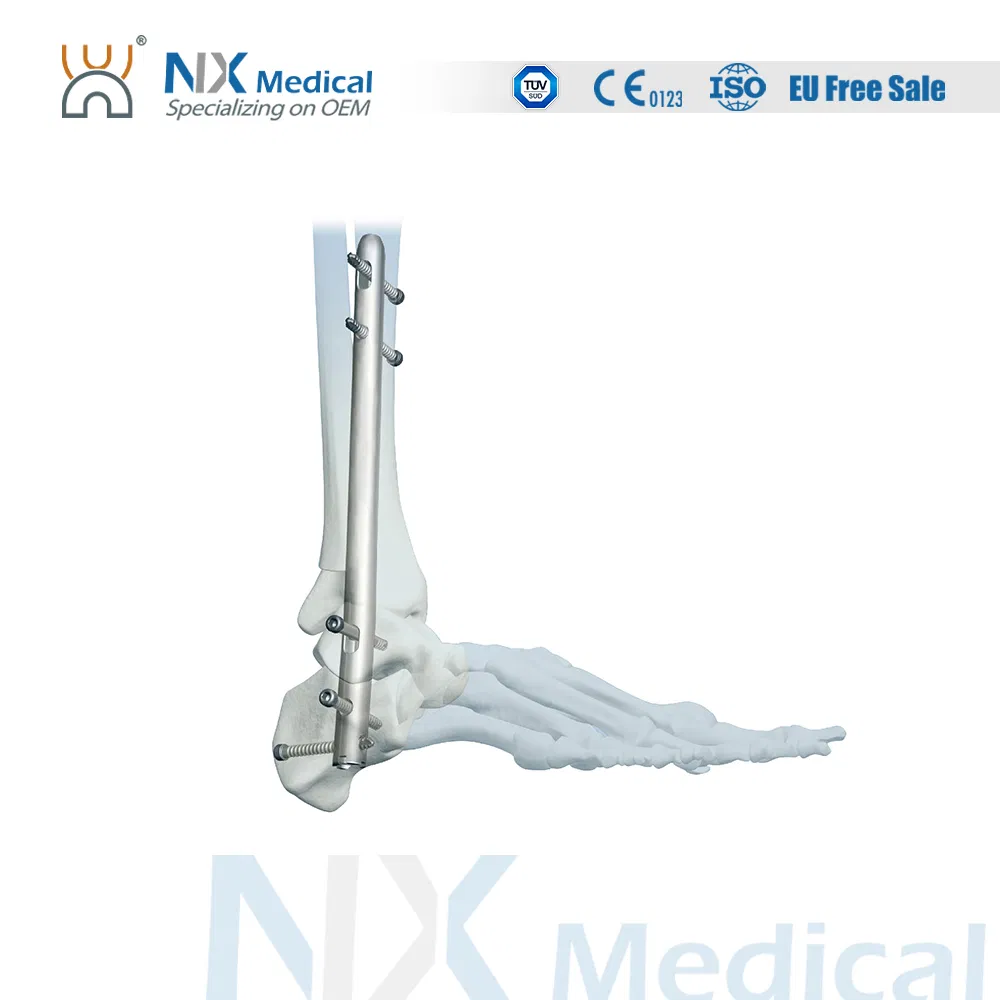
| Ankle Fusion Nail | 9 x 160/180/200/220/240 | Titanium Alloy (TA) |
| 10 x 160/180/200/220/240 | Titanium Alloy (TA) | |
| 11 x 160/180/200/220/240 | Titanium Alloy (TA) | |
| Tibial Nail Interlocking Screw (φ4.4mm) | 24 to 42 (2mm increments) | Titanium Alloy (TA) |
| End Cap | 0, 5, 10, 15 (Extension) | Titanium Alloy (TA) |

Our manufacturer is a leading orthopedic specialist located in East China, dedicated to providing comprehensive and cost-effective surgical therapeutic solutions. We offer a diverse range of trauma implants, spinal implants, and high-precision surgical instruments globally.
Committed to innovation through continuous R&D investment, our facilities utilize advanced CNC machinery to ensure the highest quality standards. All products are manufactured under strict internal quality control systems and hold international certifications including ISO13485 and TUV CE0123.