 Tandir Orthopedic Implants
Tandir Orthopedic Implants
 Tandir Orthopedic Implants
Tandir Orthopedic Implants
 Lockable Intramedullary Nail Titanium Alloy
Lockable Intramedullary Nail Titanium Alloy
 Expert PFNA Intramedullary Interlocking Femur Nail 10*220
Expert PFNA Intramedullary Interlocking Femur Nail 10*220
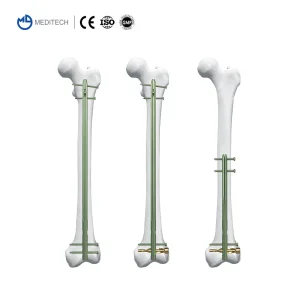 Suprapatellar Approach Tibial Intramedullary Nail
Suprapatellar Approach Tibial Intramedullary Nail
 Titanium or Ss Femoral Intramedullary Nails
Titanium or Ss Femoral Intramedullary Nails
 Orthopedic PFNA Proximal Femur Interlocking Nail
Orthopedic PFNA Proximal Femur Interlocking Nail
 Interlocking Nail in Blue Green Gold Colors
Interlocking Nail in Blue Green Gold Colors
 Multi-Screw Hole Tibial Intramedullary Nail
Multi-Screw Hole Tibial Intramedullary Nail
 Standard Intertan Intramedullary Nail for Surgery
Standard Intertan Intramedullary Nail for Surgery
The global orthopedic trauma fixation market has witnessed a paradigm shift toward minimally invasive procedures, where Intramedullary (IM) Nails have become the "gold standard" for treating long bone fractures. As leading Intramedullary Nails manufacturers & suppliers, we recognize that the industry is no longer just about stainless steel rods; it is about biomechanical harmony and rapid patient recovery.
Today, the market is driven by an aging global population prone to osteoporotic fractures and a rising number of high-energy trauma cases from road accidents and sports. From North America to the emerging markets in Southeast Asia and Africa, the demand for interlocking nail systems is skyrocketing. Hospitals are shifting from traditional plating to IM nailing because it preserves the periosteal blood supply and allows for earlier weight-bearing.
The industrial status of IM nails is characterized by intense R&D focused on Titanium Alloy (Ti6Al4V) and Cobalt-Chrome materials. Manufacturers are now integrating AI-driven design to optimize the anatomical curvature of the nails, ensuring a better fit for diverse ethnic populations. This localization of orthopedic geometry is a critical trend for global suppliers.
Modern IM nails are evolving with multi-planar locking options. This allows surgeons to stabilize complex fractures near the joints (proximal or distal) with unprecedented precision. The shift towards "Expert" systems means more screw holes at varied angles to tackle fragmented bone segments.
Research is currently heavy on antibiotic-coated intramedullary nails to prevent surgical site infections (SSI), especially in open fractures. As a forward-thinking supplier, we monitor these trends to provide solutions that reduce secondary surgery rates.
The transition from infrapatellar to suprapatellar approach for tibial nails is a game-changer. It maintains the leg in a semi-extended position, facilitating easier C-arm imaging and reducing knee pain post-surgery. Our Suprapatellar Tibial Nail systems are designed specifically for this technique.
Anodization technology allows for color-coded implants (Blue, Green, Gold). This is not just aesthetic; it assists surgical staff in quickly identifying nail diameters and types during high-pressure emergency operations.
Procurement officers in the EU, Middle East, and Latin America face the challenge of balancing cost-efficiency with high-quality standards. When sourcing from Intramedullary Nails manufacturers, global buyers prioritize CE and FDA certifications, ISO 13485 compliance, and reliable lead times.
In the post-pandemic era, supply chain resilience is paramount. Hospitals are no longer relying on a single source. They seek partners who can offer comprehensive orthopedic sets, including the nails, locking screws, and the specialized instrumentation required for implantation. Our factory in China bridges this gap by offering "One-Stop" trauma solutions.
Beijing Tandir Medical Device Co., Ltd. was established in 2022. It is a professional supplier of medical equipment, medical consumables and medical equipment, dedicated to providing high-quality solutions for the global market. Its geographical location offers convenient logistics, and it has established close cooperation with factories equipped with advanced production facilities, adhering to international quality standards and emphasizing innovation to meet the constantly changing needs of customers.
Our team combines industry expertise with technical precision, supported by partner factories featuring skilled front-line employees. From R&D to production and quality control, we collaborate closely to ensure products meet CE/FDA requirements. Customer-centric service and timely delivery are core values embedded in our operations.

As a leading Chinese orthopedic factory, we offer several strategic advantages that Western competitors often struggle to match. The convergence of industrial scale and technological maturity in China has made it the global hub for medical device manufacturing.
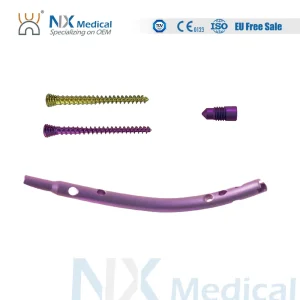 Retrograde Tibial Nail Orthopedic Implant
Retrograde Tibial Nail Orthopedic Implant
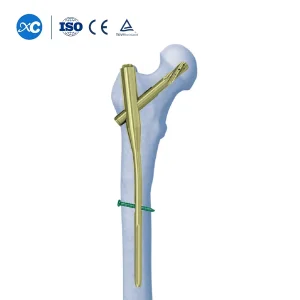 Titanium Femur Intramedullary Nail PFNA
Titanium Femur Intramedullary Nail PFNA
 Expert Tibial Proximal Interlocking Nail
Expert Tibial Proximal Interlocking Nail
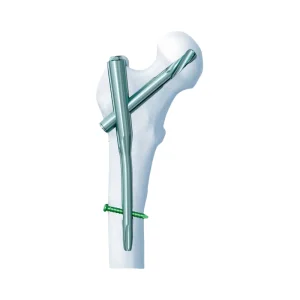 PFN Titanium Orthopedic Trauma Implants
PFN Titanium Orthopedic Trauma Implants
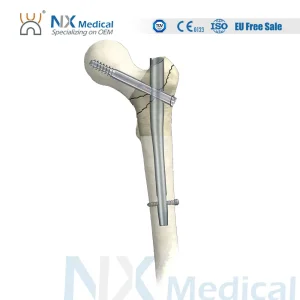 Orthopedic Trauma Titanium Interlocking Nail
Orthopedic Trauma Titanium Interlocking Nail
 Expert Humeral Proximal Interlocking Nail
Expert Humeral Proximal Interlocking Nail
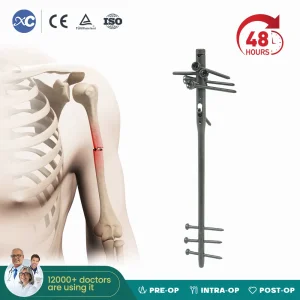 Multi-Lock Humeral Intramedullary Nail
Multi-Lock Humeral Intramedullary Nail
 Pure Titanium ISO CE Interlocking Nail
Pure Titanium ISO CE Interlocking Nail
In the realm of orthopedic trauma, the choice of material for an intramedullary nail is a critical decision that impacts the long-term success of fracture fixation. Historically, Stainless Steel 316L was the material of choice due to its high strength and relative ease of manufacturing. However, the modern industry has pivoted towards Titanium Alloys, specifically Ti-6Al-4V ELI (Extra Low Interstitial).
Why Titanium? The answer lies in the Modulus of Elasticity. Titanium’s modulus is much closer to that of human cortical bone compared to stainless steel. This reduces the effect of "stress shielding," where the implant carries too much of the load, causing the surrounding bone to weaken over time. By using titanium intramedullary nails, we promote Wolff’s Law, encouraging the bone to heal and strengthen through natural physiological loading.
Furthermore, biocompatibility is significantly higher with titanium. It forms a stable oxide layer that prevents corrosion and minimizes the release of metal ions into the body. This is particularly important for patients with metal sensitivities. As global Intramedullary Nails suppliers, we also provide Type II Anodized surfaces, which increases the fatigue strength of the nail and reduces the risk of cold-welding between the nail and the locking screws, making future removal (if necessary) much easier for the surgeon.
Mechanically, the design of the nail’s cross-section has also evolved. While solid nails were common, cannulated intramedullary nails have become the standard because they allow for the use of a guide wire, ensuring precise placement within the medullary canal. Our 11 production lines are equipped with high-precision deep-hole drilling machines to ensure the perfect centricity of these cannulations, which is vital during reamed insertion procedures.
Finally, we must consider the biomechanics of interlocking. The stability of a long bone fracture under torsion and axial compression depends entirely on the locking screws. Our systems utilize a combination of static and dynamic locking holes. Dynamic locking allows for controlled axial micro-motion, which is proven to stimulate callus formation in certain types of fractures. This level of technical sophistication is what separates a generic manufacturer from a specialized medical device partner like Beijing Tandir.