

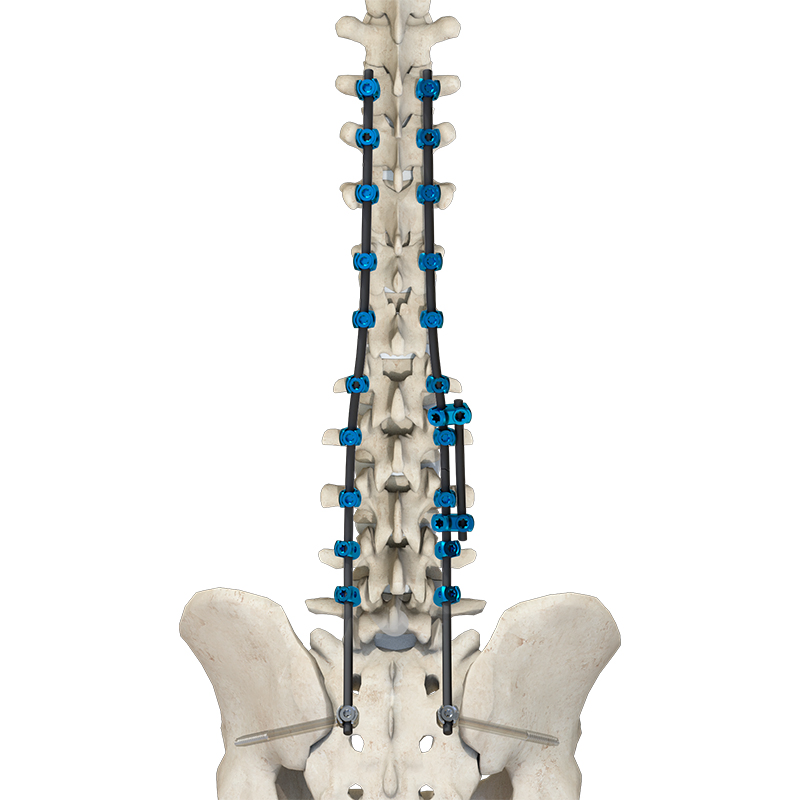
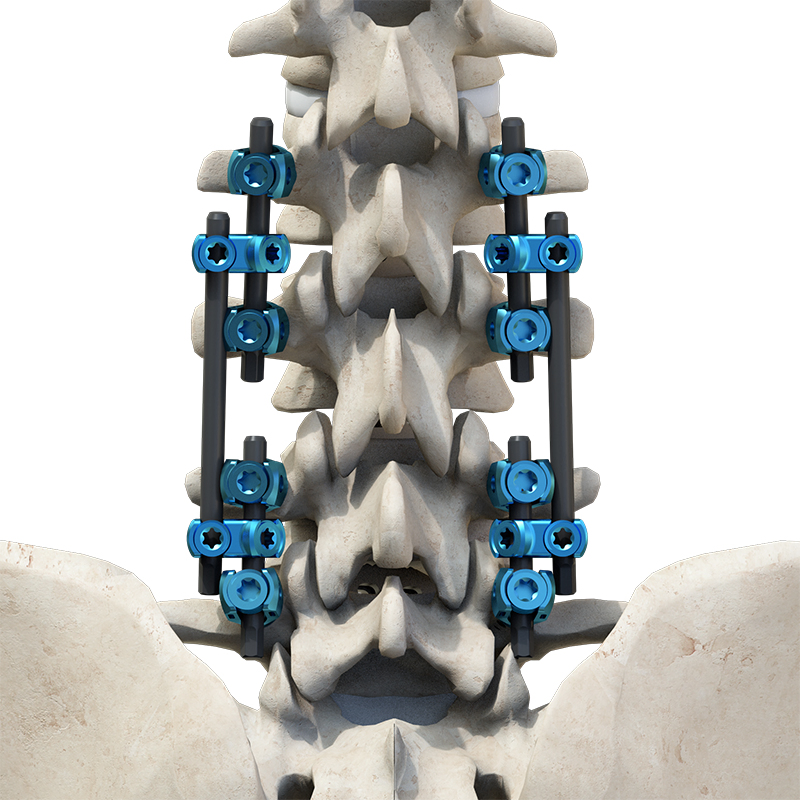
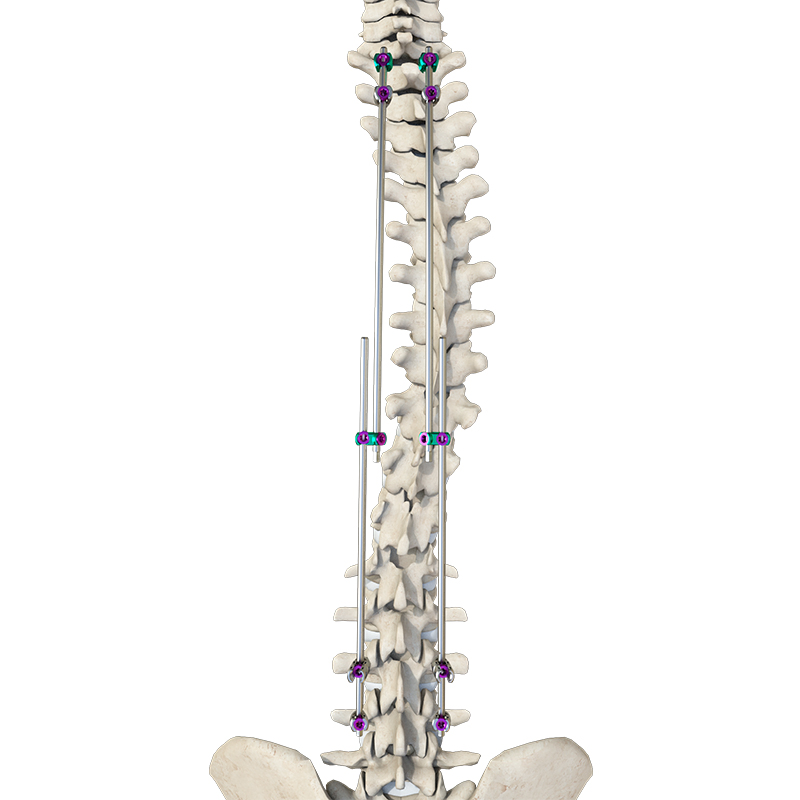
1 / 5
 Tandir Orthopedic Implants
Tandir Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |


| Model Number | Specification (mm) |
|---|---|
| 1072401 | S |
| M |

Reliable Orthopedic Solutions for Global Healthcare
As a leading manufacturer of standardized orthopedic implants and instruments, we serve global distributors and importers with excellence. Our comprehensive product range covers trauma, spine, joints, sports medicine, and CMF, all supported by precision-engineered surgical instrument sets.
With advanced in-house R&D, certified laboratories, and modern production lines, we provide scalable manufacturing solutions, including OEM/ODM services for partners requiring custom or private label implants.