Tandir Orthopedic Implants
Tandir Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 50/Piece Request Sample |
The Hook Guide Pin is an essential surgical instrument designed to facilitate accurate alignment and temporary fixation during orthopedic procedures. Its unique hooked tip provides secure engagement with bone or implants, enabling controlled positioning and stabilization throughout screw placement, plating, or drilling steps. Made from high-strength surgical stainless steel, the guide pin ensures durability, corrosion resistance, and compatibility with imaging systems for intraoperative verification.
This versatile instrument is particularly useful in procedures involving anatomical contours or hard-to-reach areas, where traditional straight guide pins may be less effective. The hook design allows surgeons to maintain alignment in complex fracture patterns or during minimally invasive approaches, improving procedural accuracy and reducing operative time.


| Product Name | Hook Guide Pin |
| Tip Design | Hook-shaped, atraumatic engagement end |
| Material | High-Strength Surgical Stainless Steel (AISI 316L) |
| Surface Finish | Polished, smooth, corrosion-resistant |
| Sterilization | Autoclavable (reusable) |
| Common Lengths | 100mm, 150mm, 200mm (Customizable) |
| Diameter Options | 1.5mm, 2.0mm, 2.5mm, 3.0mm |
| Compatibility | Cannulated systems, drilling guides, and fluoroscopy |
| Regulatory | CE Marked, ISO 13485 compliant |

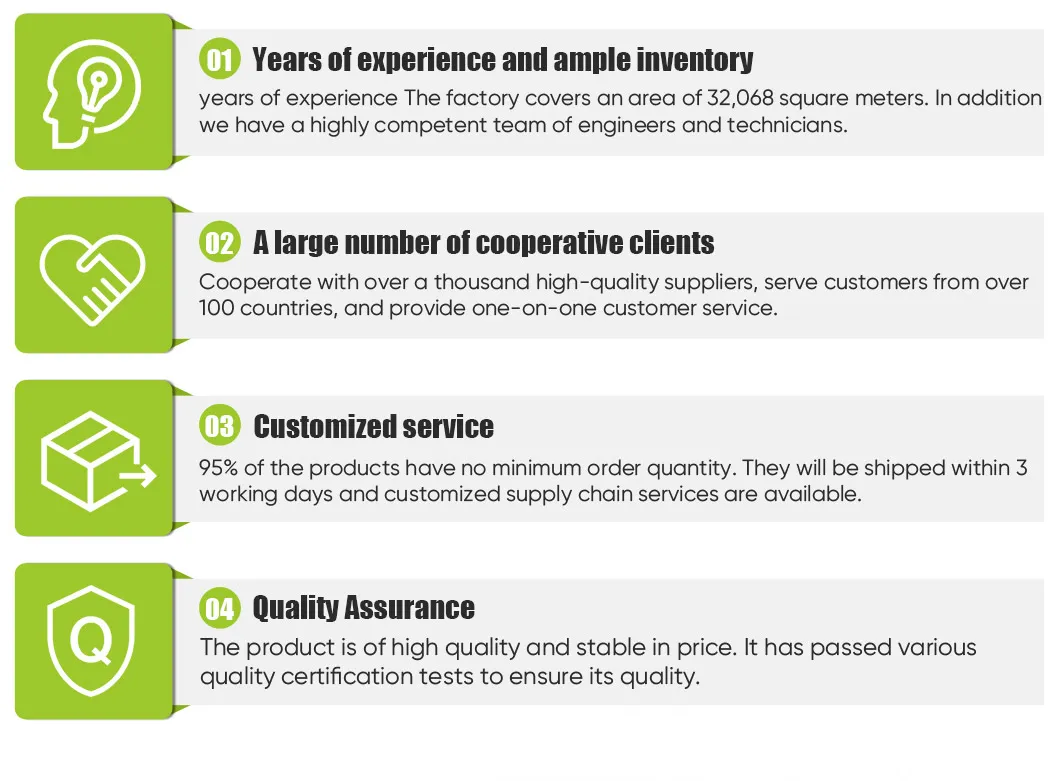
The facility is specialized in developing production and sales of orthopedic implants and instruments. With a ground space of 32,068 square meters and a strong group of engineers and technicians, the production process satisfies global requirements with advanced processing technology and testing equipment.
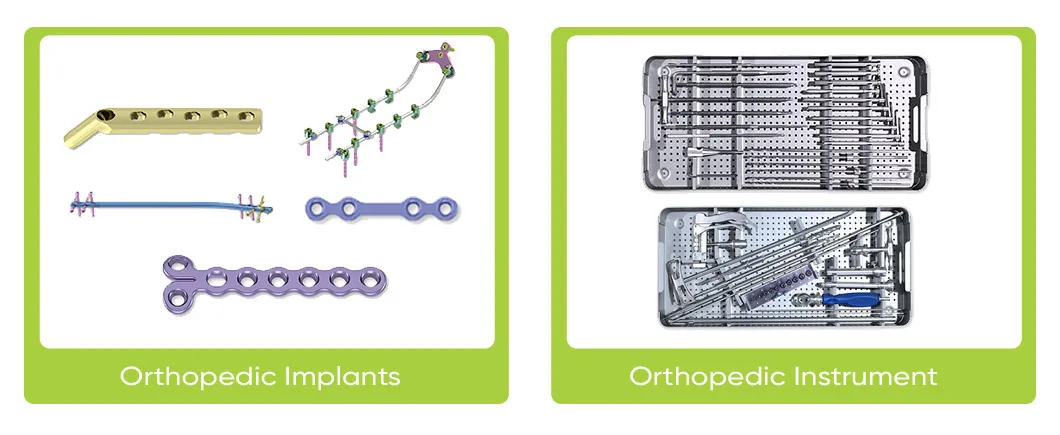
Key offerings include spinal fixation systems, interlocking intramedullary nails, metal bone plates, metal bone screws, and related operation instruments. The systems are ISO 9001 and ISO 13485 certified, with products holding CE certification.