




 Tandir Orthopedic Implants
Tandir Orthopedic Implants










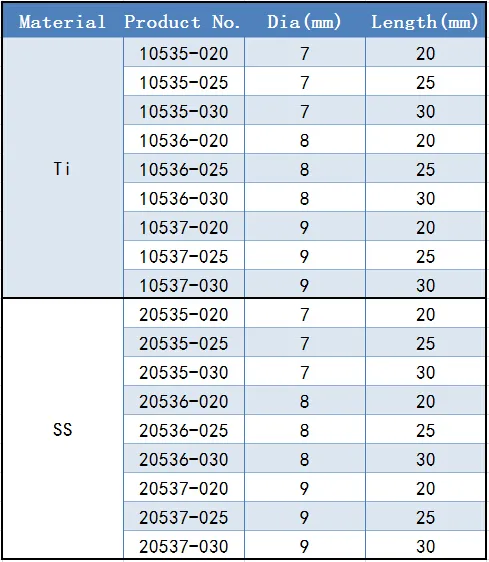
| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
As a national high-tech enterprise, R&D investment accounts for over 8% of total sales. We have pioneered revolutionary technologies globally, including the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plates. These innovations are fully endorsed by medical experts and the international market.

Our manufacturing philosophy is based on "Good faith as the base, quality above all." Operating strictly under ISO13485 and CE MDD 93/42/EEC directives, we ensure international standard quality through constant upgrading and process control.
Advanced Equipment Infrastructure:

Our orthopedic products are exported to numerous countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback from these markets confirms that our product quality consistently meets or exceeds local medical standards.