
1 / 5
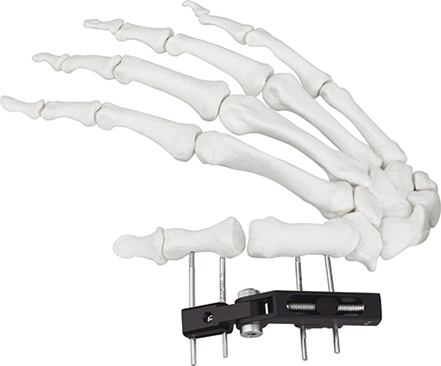
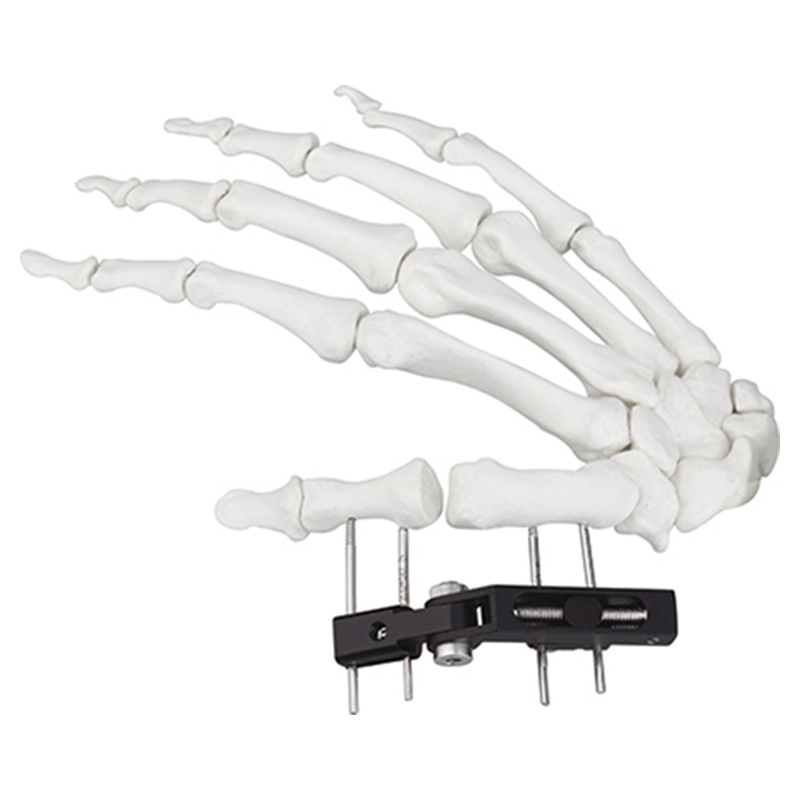
 Tandir Orthopedic Implants
Tandir Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 35/Piece Request Sample |
| Number | Product Name | Specifications | Material |
|---|---|---|---|
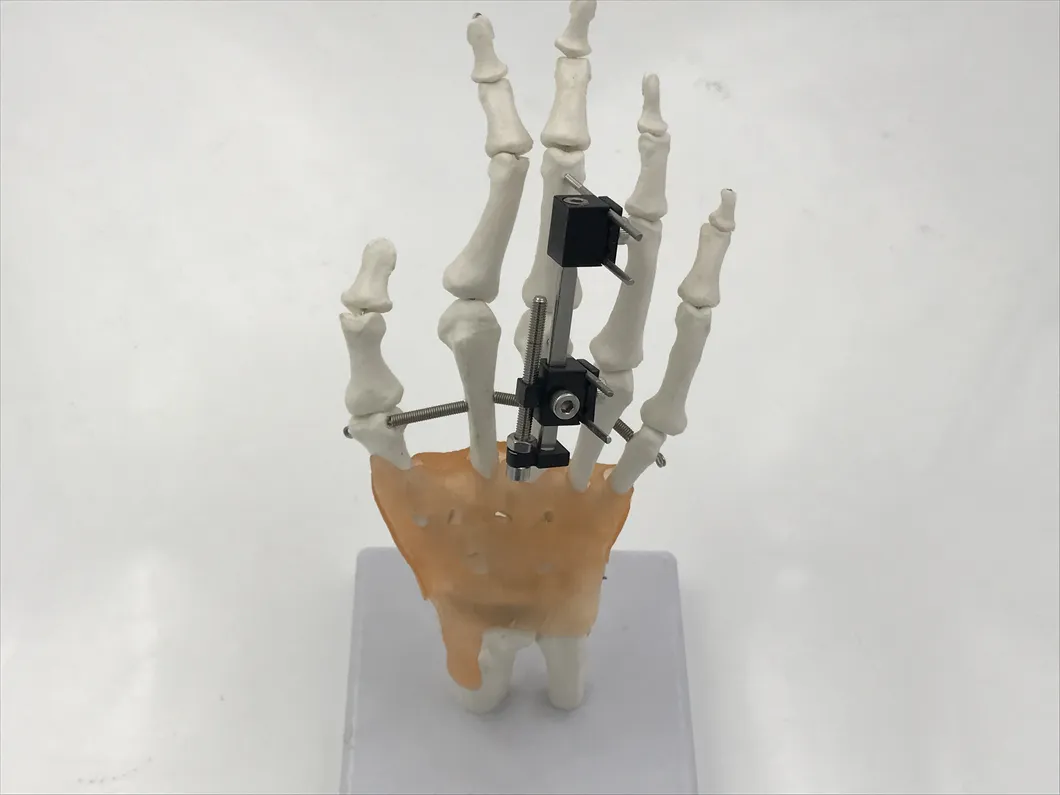
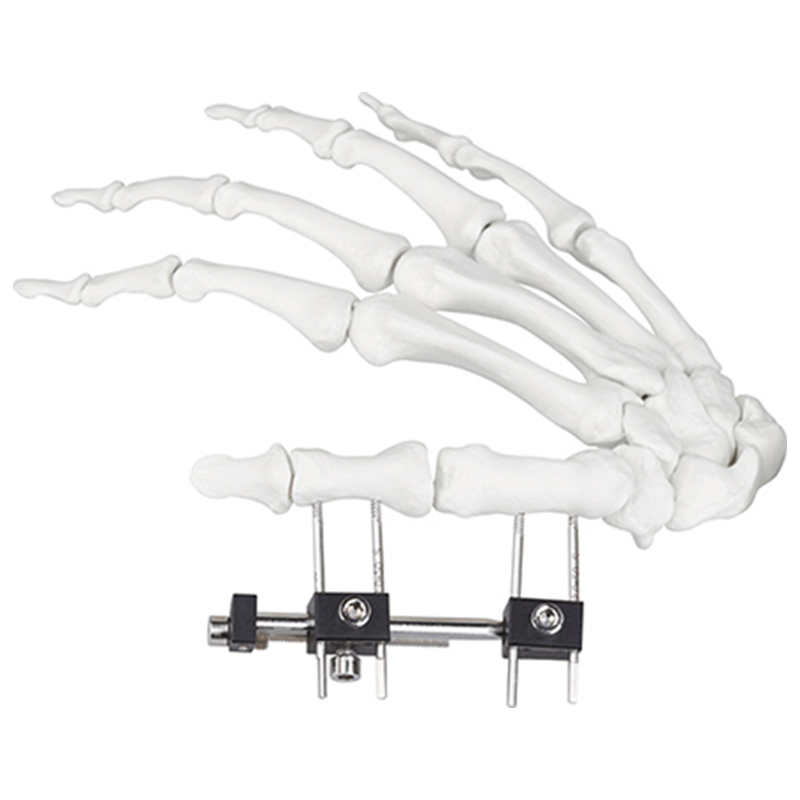
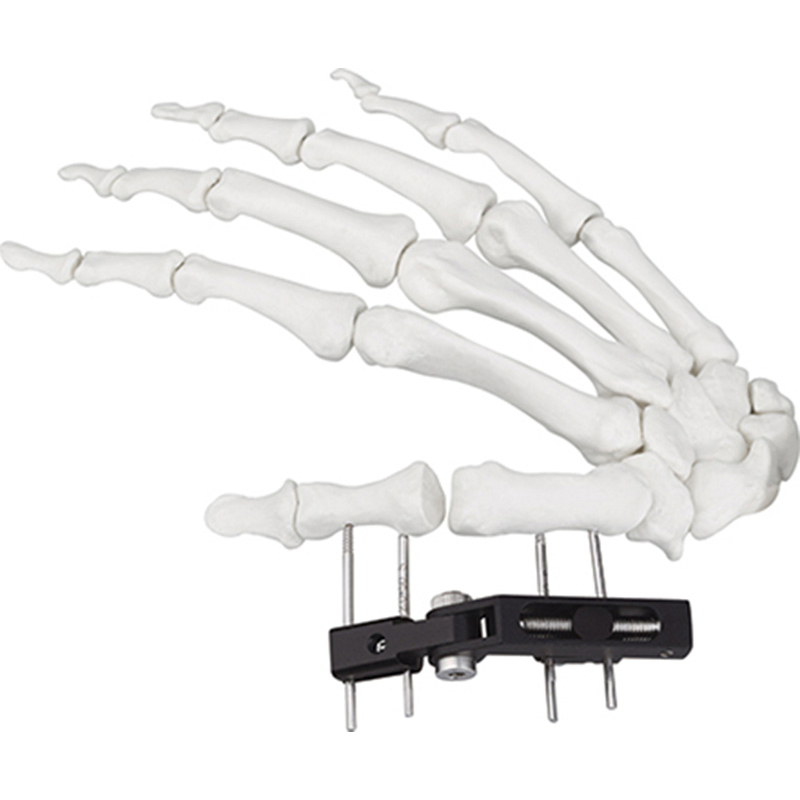
| W03 | Mini fixator universal compression extension type | Standard screw: φ2.5*50mm 4pcs; 3mm hex wrench, 2mm screwdriver | AL |