

1 / 5
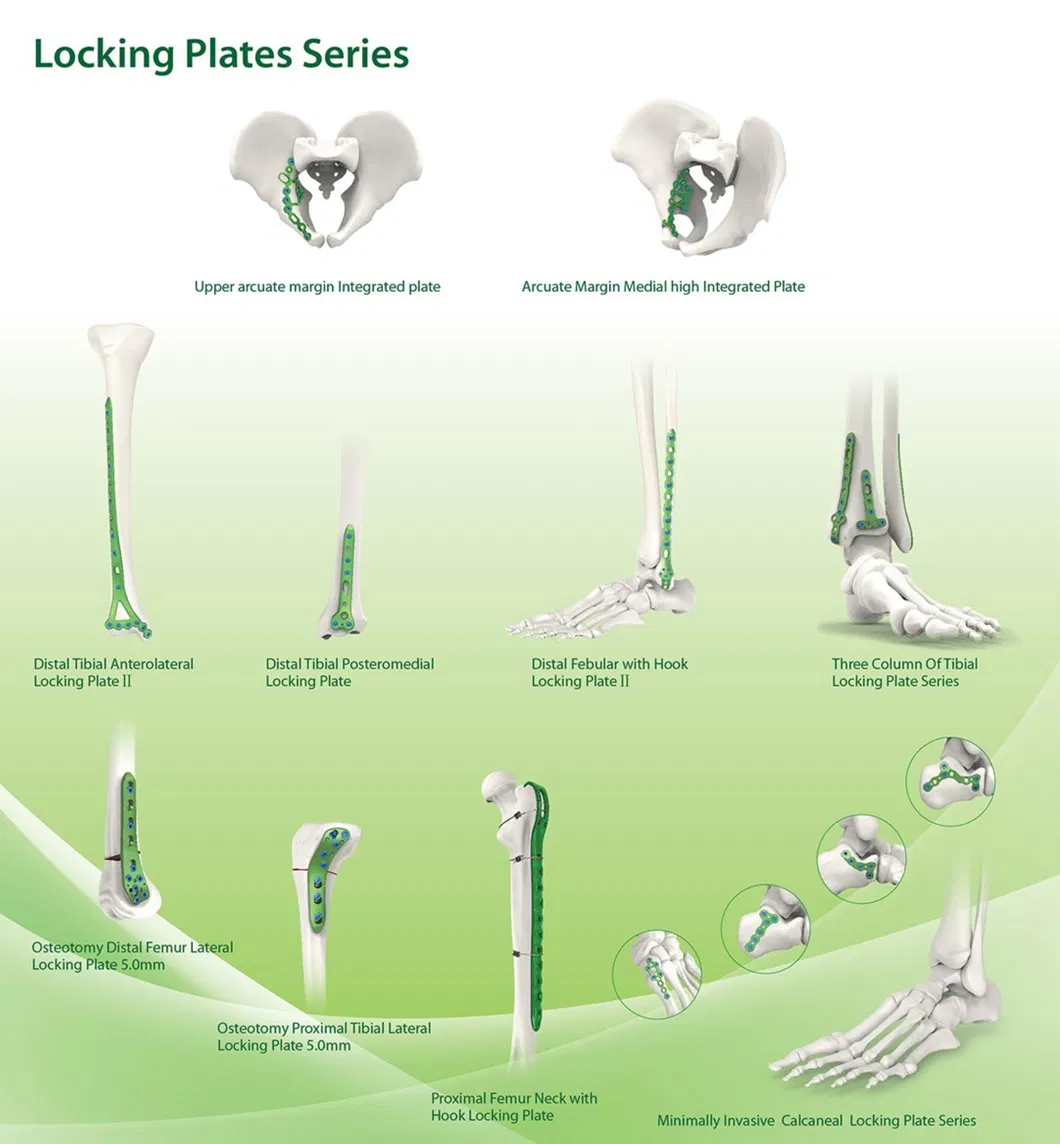
 Tandir Orthopedic Implants
Tandir Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 15/Piece Request Sample |
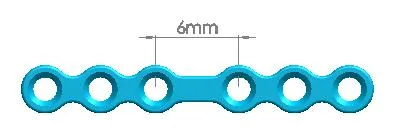
| Products Name | NO. | Specification |
|---|---|---|
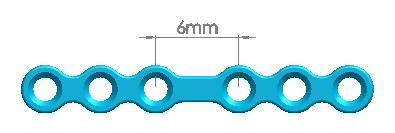
| Straight Maxillofacial Plate, Type-IV | 1500-0115 | 6 holes (Interspace:6.0mm) |
| 1500-0116 | 6 holes (Interspace:7.5mm) | |
| 1500-0117 | 6 holes (Interspace:9.0mm) | |
| 1500-0118 | 6 holes (Interspace:12mm) | |
| Use: 1.5mm Self-tapping Screw / 1.5mm Self-drilling Screw | ||