




1 / 5
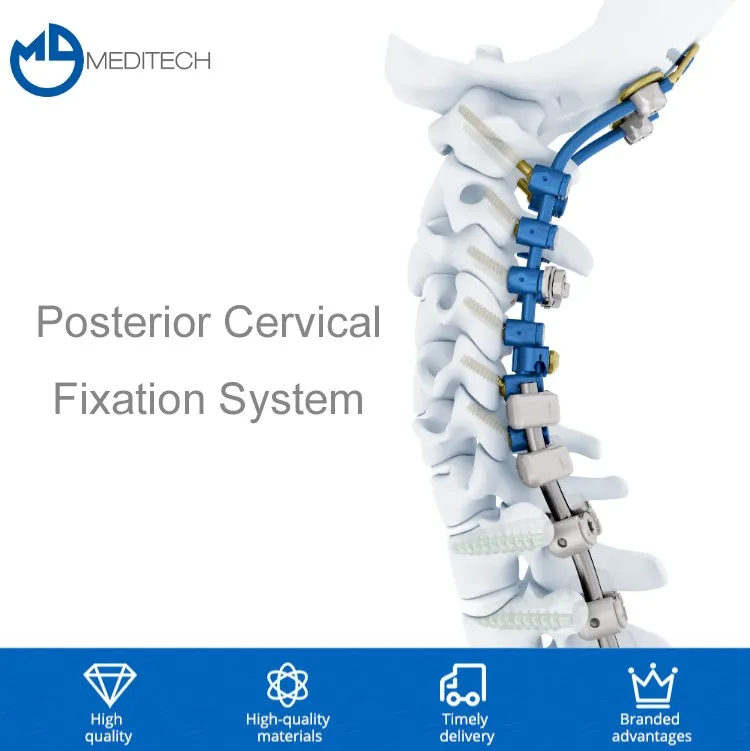
 Tandir Orthopedic Implants
Tandir Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 29/Piece Request Sample |
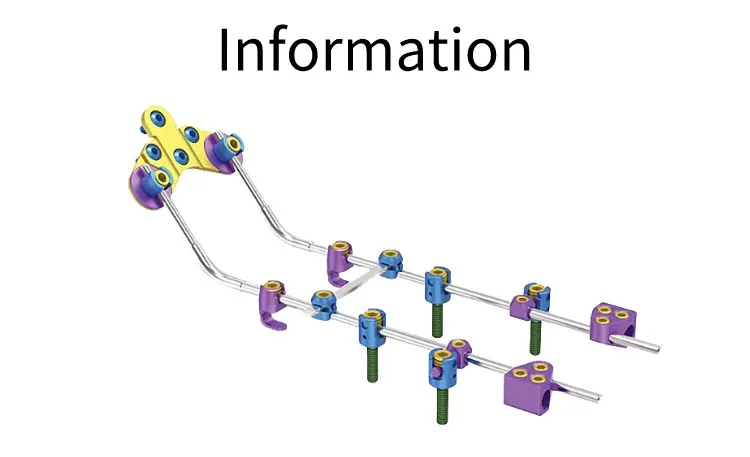
| Product | REF | Specification |
|---|---|---|
| Polyaxial Pedical Screw | 2100-1101 to 1110 | Φ3.5 × 12mm - 30mm |
| 2100-1111 to 1120 | Φ4.0 × 12mm - 30mm | |
| Occipital Plate-I | 2100-1201 | 32mm |
| 2100-1202 | 37mm | |
| 3.5mm Rod | 2100-1301 | 100mm |
| 2100-1302 | 200mm | |
| Occipital Plate-II | 2100-1401 | 4 holes |
| 2100-1402 | 5 holes | |
| 2100-1403 | 6 holes | |
| Crosslink | 2100-1501/02/03 | 35mm / 40mm / 45mm |
| Laminar Hook | 2100-1601 | Standard |
| Occipital Screws | 2100-1701 to 1704 | 4.0 * 10mm - 16mm |
| Domino Bolt | 2100-3801 | 3.5/6.0mm |









