 Tandir Orthopedic Implants
Tandir Orthopedic Implants
 Tandir Orthopedic Implants
Tandir Orthopedic Implants
Located in the heart of Europe, Luxembourg serves as a strategic distribution point. Our supply chain ensures that clinics in Luxembourg City, Esch-sur-Alzette, and Differdange receive surgical consumables with minimal lead times, ensuring that orthopedic surgeries for rotator cuff repairs and ACL reconstructions are never delayed.
Integration of bioactive coatings that promote bone ingrowth (Osteoconduction) is becoming the standard for premium Luxembourgish orthopedic solutions.
Smaller anchor footprints allow for multiple fixation points in complex glenohumeral instability cases without compromising bone integrity.
Beijing Tandir Medical Device Co., Ltd. was established in 2022. It is a professional supplier of medical equipment, medical consumables and medical equipment, dedicated to providing high-quality solutions for the global market. Its geographical location offers convenient logistics, and it has established close cooperation with factories equipped with advanced production facilities, adhering to international quality standards and emphasizing innovation to meet the constantly changing needs of customers.

Our team combines industry expertise with technical precision, supported by partner factories featuring skilled front-line employees. From R&D to production and quality control, we collaborate closely to ensure products meet CE/FDA requirements. Customer-centric service and timely delivery are core values embedded in our operations.
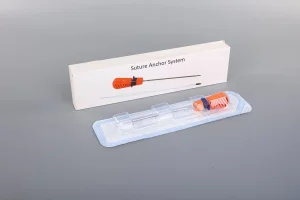
Utilized in specialized clinics across Luxembourg for middle-aged patients and athletes suffering from chronic shoulder pain and tendon tears.
Essential for the high volume of sports-related ligament injuries treated at the National Institute of Sports Medicine (INNS) in Luxembourg.
Providing titanium and PEEK solutions for complex ACL/PCL reconstructions, ensuring long-term stability for active residents.







"Providing the bridge between advanced manufacturing technology and clinical excellence in the Grand Duchy."