 Tandir Orthopedic Implants
Tandir Orthopedic Implants
 Tandir Orthopedic Implants
Tandir Orthopedic Implants
The global orthopedic market is witnessing an unprecedented surge in demand for Thoracolumbar Fixation Systems. As spinal disorders, traumatic injuries, and aging populations become more prevalent worldwide, the industry has shifted toward high-precision, biocompatible implants that ensure long-term stability and rapid patient recovery.
Current commercial trends indicate a valuation exceeding billions of dollars, with North America, Europe, and the Asia-Pacific region leading the consumption. In this competitive landscape, China has emerged as a powerhouse of manufacturing excellence, combining cost-efficiency with rigorous international quality standards like ISO 13485, CE, and FDA compliance.
Distributors and healthcare providers are no longer just looking for a "supplier"—they are seeking a partner that understands the biomechanical complexities of the spine. From titanium alloy pedicle screws to cobalt-chromium-molybdenum (CoCrMo) rod systems, the material science behind these devices is what defines clinical success.

Supplying Tier-1 hospitals and surgical centers across 50+ countries with reliable spinal implants.
Continuous investment in biomechanical testing and material innovation to reduce post-op complications.
Full documentation support for medical device registration in various international jurisdictions.
The transition from open surgery to MIS techniques is the most significant trend. This requires specialized cannulated screws and percutaneous delivery systems that reduce tissue damage and blood loss.
Additive manufacturing allows for the creation of porous structures that mimic bone biology, enhancing osseointegration. While standard systems remain the backbone of the industry, patient-specific implants are gaining traction.
The integration of spinal fixation with AI-guided surgical navigation and robotic assistance ensures sub-millimeter accuracy, significantly reducing the risk of neurological injury during screw placement.
Our systems are compatible with modern navigation and robotic systems.
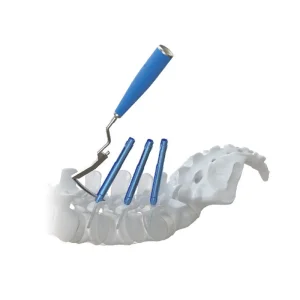
Our Minimally Invasive Surgery (MIS) pedicle screw system represents the pinnacle of current spinal engineering. Designed for percutaneous insertion, it minimizes surgical trauma while providing the same structural integrity as traditional systems.
Hospitals and procurement agencies worldwide have distinct needs based on their local healthcare infrastructure. Understanding these is key to our success as an exporter.
Trauma Fixation: High-energy fractures requiring rapid and rigid stabilization.
Degenerative Correction: Treating scoliosis or kyphosis with multi-level fixation.
Oncology: Supporting the vertebral column after tumor resection.
Developed Markets: Demand for premium CoCrMo rods and MIS sets.
Emerging Markets: Focus on durable, cost-effective titanium systems with comprehensive instrument sets for various surgical environments.
Procurement teams prioritize Lead Time and After-sales technical training. Our proximity to Beijing's logistics hubs ensures that life-saving implants reach their destination without delay.
Established in 2022, Beijing Tandir Medical Device Co., Ltd. has quickly become a cornerstone of the medical equipment industry. Our mission is to bridge the gap between advanced engineering and accessible healthcare.
Operating out of Beijing, we benefit from a rich ecosystem of medical innovation and convenient global logistics. Our partnership with state-of-the-art factories allows us to control the entire production cycle—from the raw titanium bar to the final sterile-packed implant.
Our team of over 21+ Innovation Experts works tirelessly to ensure that every screw, plate, and rod meets the exacting standards required by surgeons in the operating theater.

Beyond the spine, our expertise extends to complex fracture fixation. Our external fixators are engineered for rapid deployment in trauma centers, providing stable reduction of fractures when internal fixation is not immediately feasible.
Quality in the medical field is non-negotiable. At Beijing Tandir, we utilize partner factories equipped with Swiss-type CNC lathes and 5-axis machining centers. This level of precision ensures that thread tolerances are kept within microns, preventing "cold welding" between the screw and the locking cap.
Every batch of Thoracolumbar Fixation Systems undergoes rigorous testing:
Our commitment to CE/FDA requirements means that every product we export is backed by a trail of documentation and quality assurance that distributors can rely on.
✅ ISO 13485 Certified Facilities
✅ 100% Visual Inspection
✅ Ultrasonic Cleaning & Passivation
✅ Class 100,000 Cleanroom Packaging
Beijing Tandir Medical Device Co., Ltd. is dedicated to providing high-quality solutions for the global market. Our team combines industry expertise with technical precision, supported by partner factories featuring skilled front-line employees. From R&D to production and quality control, we collaborate closely to ensure products meet clinical needs. Customer-centric service and timely delivery are core values embedded in our operations.