Tandir Orthopedic Implants
Tandir Orthopedic Implants
 Tandir Orthopedic Implants
Tandir Orthopedic Implants
The global Intramedullary (IM) Nails market is witnessing unprecedented growth, driven by a rising geriatric population and a corresponding increase in osteoporotic fractures. IM nailing has become the "gold standard" for the treatment of long bone fractures, particularly in the femur, tibia, and humerus. Unlike traditional plate fixation, intramedullary nails offer biological fixation, preserving the periosteal blood supply and allowing for early weight-bearing, which is critical for patient rehabilitation.
From a commercial perspective, the demand is shifting towards high-strength, biocompatible materials such as Titanium Alloy (Ti6Al4V). Modern trauma centers in North America, Europe, and the Asia-Pacific region are increasingly seeking "Expert" nail systems that provide multi-directional locking options to handle complex comminuted fractures. This has created a surge in procurement needs for specialized kits like the PFNA (Proximal Femoral Nail Antirotation) and Intertan systems.
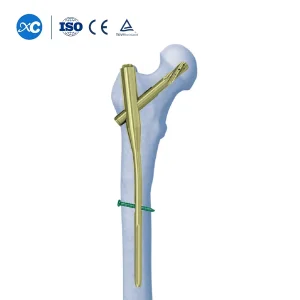
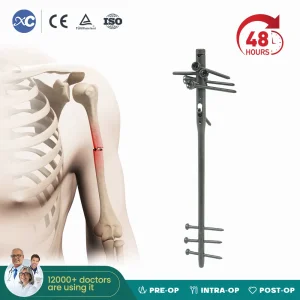
Our flagship PFNA (Proximal Femoral Nail Antirotation) is engineered for the most challenging intertrochanteric fractures. The helical blade design provides significantly higher resistance to "cut-out" compared to traditional lag screws, making it the ideal choice for osteoporotic bone. As a leading China Intramedullary Nails Manufacturer, we ensure every nail undergoes rigorous fatigue testing.
View Product DetailsOffering CE and ISO-certified implants at a fraction of the cost of Western competitors without compromising on material purity.
Our rapid prototyping allows us to iterate designs based on surgeon feedback faster than traditional industry giants.
Located in China's medical hub, we guarantee stable lead times even during global logistics fluctuations.
Customized branding and sterile packaging options tailored for local regulatory requirements in your region.
In aging societies like Japan, Germany, and Italy, the demand for PFNA nails is skyrocketing. Our products are designed to support minimally invasive surgery (MIS), reducing blood loss and operative time, which is vital for elderly patients with comorbidities.
In regions with high rates of road traffic accidents (RTA), such as Southeast Asia and Latin America, our Tibial and Femoral Interlocking Nails provide a robust and cost-effective solution for large-scale hospital tenders. We provide full instrument sets to ensure even remote clinics can perform successful surgeries.

Beijing Tandir Medical Device Co., Ltd. was established in 2022. It is a professional supplier of medical equipment, medical consumables and medical equipment, dedicated to providing high-quality solutions for the global market. Its geographical location offers convenient logistics, and it has established close cooperation with factories equipped with advanced production facilities, adhering to international quality standards and emphasizing innovation to meet the constantly changing needs of customers.
Our team combines industry expertise with technical precision, supported by partner factories featuring skilled front-line employees. From R&D to production and quality control, we collaborate closely to ensure products meet CE/FDA requirements. Customer-centric service and timely delivery are core values embedded in our operations.
The Xc Medico Titanium Interlocking Humerus Nail represents the pinnacle of upper limb fracture fixation. Featuring a unique multi-lock design, it allows for superior stability in proximal and distal humeral fractures, preventing rotation and promoting faster primary bone healing.
Explore Humeral Nails